 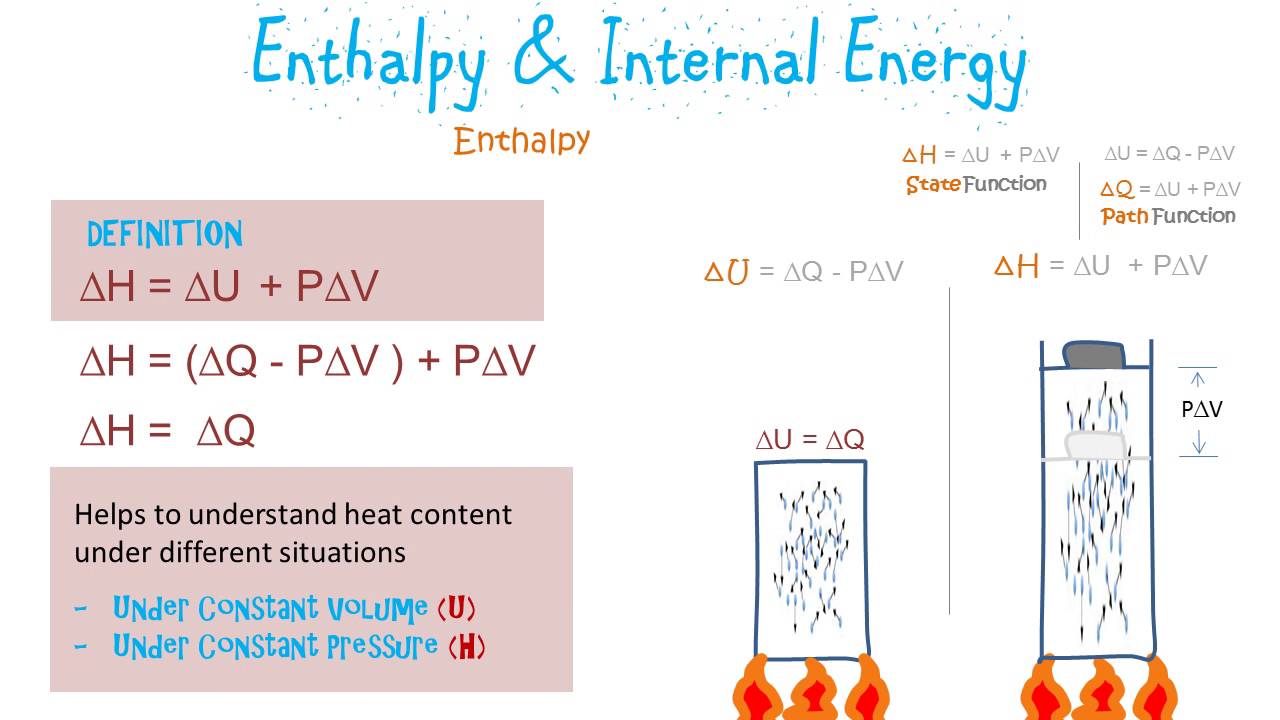
And, since they are specified exactly by my state variables $V$ and $T$, $dP$ and $dV$ are exact differentials. This makes $P = P(S, T)$ and also $V = V(S, T)$. Now I exercise my freedom, and pick $S$ and $T$ as my independent variables. Since I have two equations and four "unknown"s, it means I can choose any two of them as independent variables, and the other two will be specified by the equations. This is as far as you can deduce using thermodynamics of an ideal gas only.)įor a non-ideal gas, the equations deviate from these forms, but a gas in equilibrium always has a state equation like $PV = NkT$, and there is always a "proper" expression for $S$. $df = \big( \frac$ ($c_v$ is the specific heat of the gas at constant volume, and $\Phi$ is a gas-dependent constant. Value of the path function will be different if a different path is taken to reach the final state.įor example entropy, mass, temperature, volume, etc.For any function of multiple variables, say, $f(x, y, z, \ldots)$ you can always write the following: Value of state function remains the same regardless of the path or steps involved to reach that value. They need multiple integrals and limits of integration for integrating the property of the system. It can be integrated using values of the initial and final state of a system. It is a property whose value depends on the path followed to reach that value. It refers to a property whose value does not depend on the path followed to reach that value. Difference between state function and path function State Function A path function has variable values based on the route. 
So, we can see the work done by him depends on the path followed, so, work is a path function. In this second case, he takes the same weight from point A to point B but this time he travels 3 km, so in this case, the work done by him will be more, however, he takes the weight to the same destination. For example, a person carries a 10kg weight from point A to point B after travelling a distance of 2 km and the work done by him is W. Path function is a thermodynamic property whose value depends on the path followed to attain that value. Let us take a simple example to understand path function So, work is not a state function as we cannot say that a system will have a specific amount of work at a specific state. If different paths are chosen to reach from one point to another point, the work done will be different however you reach the same point in each case. The path functions depend on the path taken or covered between two (initial and final) states. Path function in ThermodynamicsĪ thermodynamic property that depends on the path between the initial and final state is known as the path function. So, potential energy is a state function, not a path function. So, the potential energy depends on the initial and final heights irrespective of the path followed to reach the top floor. Now if he takes a different route to reach the top floor such as stairs even then the potential energy will be mgh2 as he reaches the same height in this case. Now he reaches the top floor by lift then his potential energy becomes mgh2 where m (mass) and gravity (g) are the same only height changes. His potential energy is mgh where m is the mass of the person, g is gravity and h1 is the height of the first floor from the ground. Let us take a simple example to understand state function Ī person standing on the first floor of a building. The state functions depend on the values of different parameters at a particular time, it does not depend on how these values are obtained or irrespective of the path taken. A change in their values depends on their initial and final state. Similarly, the thermodynamics properties like internal energy (U), enthalpy (H), entropy (S), etc., are state functions. 
For example, density is a state function, it does not depend on how a substance is obtained. 
Properties that depend on state include pressure, temperature, amount of substance, etc. State function in ThermodynamicsĪ thermodynamic property that depends only on the initial and final state and does not depend on the path taken to reach the final state or value is known as the state function or point function. State and path functions both are thermodynamics terms that are used to name these properties. These quantities that can identify the state of a function are called properties. The state of a system is determined with the help of some measurable quantities like volume, temperature, density, pressure, etc. Next → ← prev State and Path function in Thermodynamics 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
